Reverse Thiazine Kinase Inhibitors
Summary
The National Cancer Institute (NCI) seeks research co-development partners and/or licensees for a class of novel aplithianine-derived small molecule analogs that compete with ATP for binding on a range of clinically relevant kinases including:
- Oncogenic gene fusion DNAJB1-PRKACA (PKADJ)
- Wild type protein kinase A (PKA)
- Protein kinase G (PKG)
- Ccdc2-like kinases (CLK) 1 & 2
- DYRK family of kinases
Description of Technology
In 2022, the NCI Molecular Targets Program (MTP) completed a screen of ~150,000 pre-fractionated natural products from the NCI Program for Natural Product Discovery (NPNPD). From this screen, a class of active compounds, named Aplithianines A & B (isolated from the marine organism Aplidium sp.) showed broad potential applicability to numerous kinases of importance including but not limited to:
- Oncogenic gene fusion DNAJB1-PRKACA (PKADJ)
- Implicated in an ultra-rare adolescent liver cancer
- Wild type protein kinase A (PKA)
- Implicated in Cushing’s Disease
- Protein kinase G (PKG)
- Potential treatment of malaria
- Ccdc2-like kinases (CLK) 1 & 2
- Implicated in gastric cancer
- DYRK family of kinases
- Implicated in gastric or colon cancer as well as infections caused by a protozoa or parasites
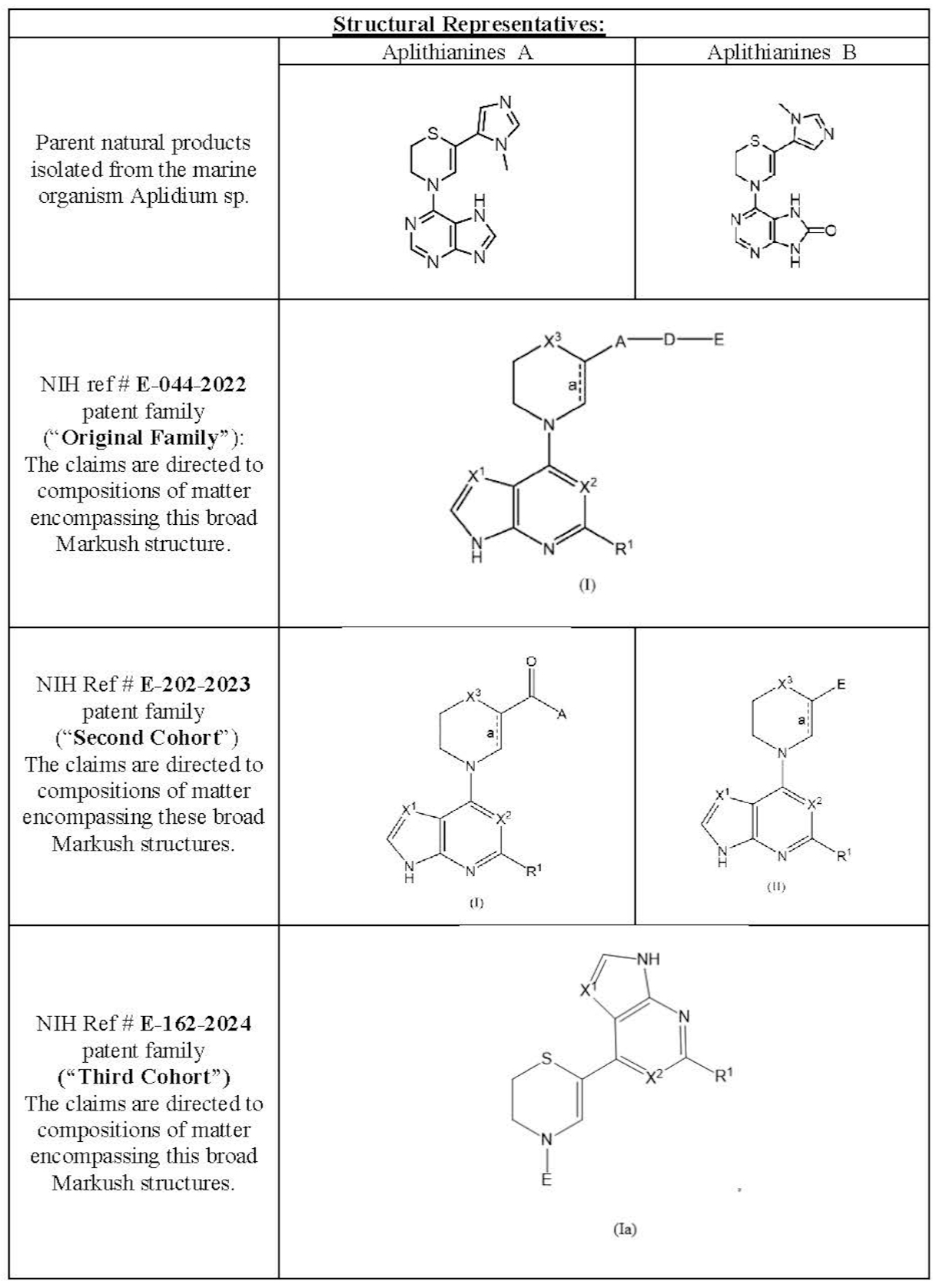
This Technology (NIH Ref # E-162-2024) describes a Third Cohort of compounds, namely the Reverse Thiazine Kinase inhibitors – structurally related to, but patentably distinct from – compounds described in the patent filings for the Original Family and Second Cohort. This Third Cohort of reverse thiazine compounds were created based on the initial activity determination and structural interactions for the aplithianine kinase inhibitors (described in the Original Family) and the DNAJ-PKA fusion protein. These Third Cohort compounds are a new structural class of Ser/Thr kinase (i.e. PKA) inhibitors related to the aplithianine class of kinase inhibitors but differ in both their chemical structures and mode of binding into the ATP binding pocket of kinases. They show potent and improved biochemical inhibition, reduction of the phosphorylation of the PKA substrate CREB, and the ability to reduce the viability of cancer cells. Computational modeling predicted enhanced binding affinity of these reverse thiazines with the DNAJ-PKA fusion protein. Consistent with this hypothesis-driven modeling effort, the reverse thiazines show improved potency and cellular activity over the compounds described in the Original Family and Second Cohort patent families
Original Cohort of compounds, including the pharmaceutical compositions of the natural products Aplithianine A and Aplithianine B, as well as a range of synthetic derivatives are described in NIH Technology Ref # E-044-2022.
The Second Cohort of compounds are covered in (NIH Ref # E-202-2023). This Second Cohort comprises the same chemical scaffold of the broadest generic formula in the Original Family but represents a patentably distinct, subgenus formula. This Second Cohort of compounds shares the same chemical scaffold as aplithianine but have been optimized through extensive medicinal chemistry efforts to increase binding affinity to the oncogenic fusion kinase called DNAJB1-PRKACA (PKADJ) and its wild-type counterpart protein kinase A (PKA).
The specificity of several of the compounds have been examined in kinase panels to demonstrate that, while applicable to a range of kinases, they are not promiscuous kinase inhibitors. The subject kinase inhibitors have broad potential commercial applicability’s for cancer, immune suppression, preventing organ rejection, treating diabetic neuropathic pain, malaria, or protozoa infection. To date, there are no approved therapeutics targeting DNAJB1-PRKCA, an oncogenic gene fusion is ubiquitously and exclusively detected in the tumors of patients with ultra-rare fibrolamellar hepatocellular carcinoma FLHCC.
The NCI seeks licensing and/or co-development research collaborations for the future development of Kinase Inhibitory Aplithianines targeting DNAJB1-PRKACA (PKADJ), PKA, PKG, CLK, and/or DYRK.
Potential Commercial Applications
- Gastric Cancer
- Ultra-rare, adolescent liver cancer
- Solid cancers susceptible to kinase inhibitors
- Cushing’s Disease
- Transplantation
- Diabetic neuropathic pain
- Malaria
- Protozoa infection
Competitive Advantages
- Applicability to numerous clinically relevant kinases, including:
- Oncogenic gene fusion DNAJB1-PRKACA (PKADJ)
- Wild type protein kinase A (PKA)
- Protein kinase G (PKG)
- Ccdc2-like kinases (CLK) 1 & 2
- DYRK family of kinases
- Applicable to range of kinases, but are not promiscuous kinase inhibitors
- Broad potential commercial applicability for several blockbuster indications, including: cancer, immune suppression, transplantation, diabetic neuropathic pain, malaria, and protozoa infection
- No approved therapeutics targeting DNAJB1-PRKCA
